Phase I/II Trial for Bismab-A
Two clinical trials in acute myeloid leukemia with the Bi-213 based drug named Bismab-A have been completed with promising results. They were both conducted by Dr. Joseph Jurcic at Memorial Sloan Kettering Cancer Center. Further development of Bismab-A will not be pursued by API at this time for supply, logistics and cost reasons. Bismuth-213 is made from actinium-225 in a process that has to be performed at a hospital using high quantities of actinium 225. This leads to severe supply limitations, high costs and special training requirements. By developing a new technology for utilizing actinium 225 directly in our drugs, we believe we have improved the drugs’ potency, cost margin and ease of use.
This new technology for using actinium 225 directly in our drug candidates enabled us to start a Phase I AML trial with the second generation drug named Actimab-A. It is an ongoing Phase I AML trial at MSKCC under physician IND conducted by Dr. Jurcic with a single dose of Actimab-A. This trial has already enrolled 18 patients and is providing safety data and an estimate of the phase II dose necessary for future Company sponsored trials in this disease.
When actinium 225 was used in the drug construct as a single agent in a single dose, it resulted in elimination of leukemia cells in 3 patients, and two patients were able to receive additional therapies they could have not received otherwise, which lead to a long term survival and possibly a cure in those cases. In contrast, when bismuth 213 was used in the drug construct, no elimination of leukemia cells had been seen with a single agent single dose approach. At the same time, the use of actinium 225 resulted in cost of goods sold on the per patient basis decrease from ~$ 70,000 to ~$4,000 with a potential of significant further decrease as API continues development of its patent filed new manufacturing technology.
Both Bismab-A and Actimab-A use the same monoclonal antibody as a targeting agent but they rely on Ac-225 and Bi-213 respectively as payloads.
Phase I/II Trial for Bismab-A
This Phase I/II trial has been completed which makes Bismab-A clinically the most advanced API drug. Despite the success of the trial, a comparison to the second generation agent, Actimab-A which has far superior potency at lower dosing levels, supply and logistics advantages and far lower cost has led us to pursue the development of the second generation agent while putting the first drug on hold. However, because of Bismab-A’s similarity to Actimab-A, API believes results of the Bismab-A Phase I/II trial provide an important indication of the safety and efficacy of Actimab-A.
Eligibility criteria for the Bismab-A trial were similar to those for the Phase I single dose trial with Actimab-A:
- Poor prognosis AML patients over the age of 60 ineligible for other treatments
- Patients with relapsed or refractory AML
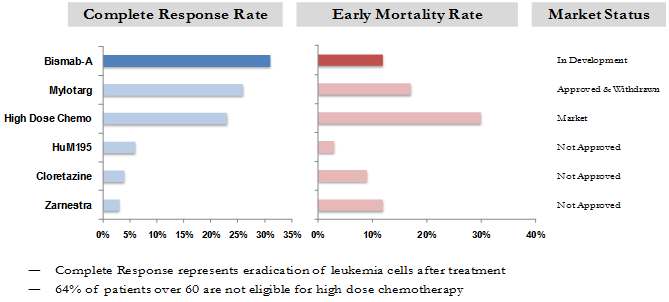
Bismab-A has been administered to almost 50 patients to date. It has maintained a low side effect record and shown clear indications of efficacy. Therapeutic dose has been established at 1 mCi/kg (mCi-milliCurie; kg- kilogram) – 1.25 mCi/kg. Twenty patients were treated at that dose range and results showed comparable efficacy with early therapy related mortality significantly lower than among the patients with similar characteristics treated by approved therapies and other therapies in clinical development.
Comparison of safety and efficacy data of Bismab-A to approved products and others under development in similar patient populations (relapsed/poor cytogenetics/secondary AML patients over 60 years of age) is presented in Figure 5 below. The data indicate that the Company’s lead product candidate has efficacy similar to approved products and superior to pipeline products, but at the same time has a better side effect profile than approved products and therefore could provide treatment to patients currently ineligible for treatment with approved products.
Comparative Safety and Efficacy of Bismab-AML at 1 mCi/kg – 1.25 mCi/kg dose